
Previous
Liver Health May Be Reflected in the Fingernails

Next
What Does Our Tongue Tell Us About Our Liver?
Knee Osteoarthritis and Limbrel: A Liver-Friendly Solution?
Medical foods are a relatively new frontier in healthcare. A medical food used for knee osteoarthritis seems safe enough at first glance, but may pose a hazard to those with liver disease.
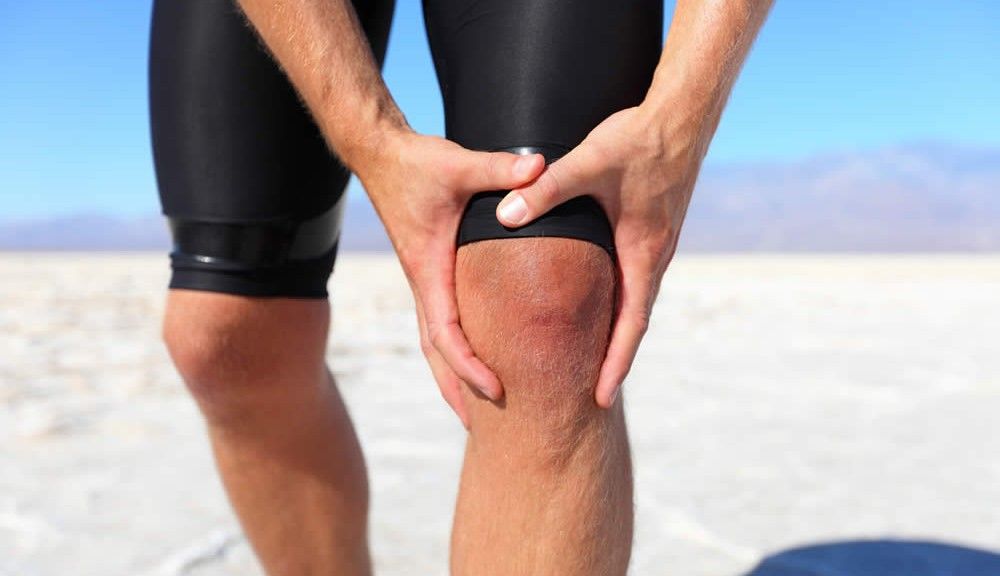
By far the most common type of arthritis in adults, more than 10 million Americans have knee osteoarthritis. There are several types of interventions used to ease this kind of knee pain, but anti-inflammatory medications are typically regarded as the standard. Since pharmaceutical anti-inflammatories can have a long list of side effects, a new, supposedly safer type of pain reliever has emerged. Belonging to the medical foods category, Limbrel is a popular, physician-prescribed supplement designed to help ease the pain and damage of knee osteoarthritis. However, it’s categorization as a ‘medical food’ and not a ‘drug’ can bring a false sense of safety – especially for those who have a compromised liver.
About Knee Osteoarthritis
Considered to be the type of arthritis associated with wear and tear, osteoarthritis is a degeneration of the joint. Frequently associated with metabolic and inflammatory anomalies, osteoarthritis symptoms typically include the following:
• Joint discomfort and pain
• Stiffness and limited mobility
• Joint swelling and tenderness
Since the symptoms of knee osteoarthritis can escalate as the disease progresses, most physicians are proactive in preventing its advancement. Knee osteoarthritis cannot be cured; however, there are treatments that help manage the symptoms and prevent further cartilage deterioration. The most common treatments include:
• Medications – Over-the-counter or prescription pain relievers and anti-inflammatories can deliver significant relief.
• Exercise– Since it increases the quantity of joint fluid, moving the joint can help protect against further deterioration.
• Physical Therapy and Acupuncture – Both of these modalities increase blood and fluid circulation around the joint, which facilitates the healing process.
• Weight Loss – For those who are overweight, losing weight eases the knees’ burden.
• Injections – Steroids injected into the joint can quell inflammation, and injecting hyaluronic acid may help lubricate the joint.
• Surgery – Arthroscopic surgery could help repair or remove irritants and smooth rough edges of the knee joint. A knee replacement is usually the last resort.
Other ways of managing knee arthritis may include hot and cold therapy, topical creams, knee supports, glucosomanine/chondroitin supplements and medical foods.
What Is a Medical Food?
Coined as a legitimate type of treatment in the early 90s, medical foods lie in the center of the prescription drug/natural supplement continuum. Bypassing the rigorous clinical trials required of a new medication and capitalizing on the growing recognition of dietary therapy’s efficacy, medical foods fill a unique niche in western medical practices.
The term medical food, as defined in section 5(b) of the Orphan Drug Act (21 U.S.C. 360ee (b) (3)) is “a food which is formulated to be consumed or administered enterally under the supervision of a physician and which is intended for the specific dietary management of a disease or condition for which distinctive nutritional requirements, based on recognized scientific principles, are established by medical evaluation.” Medical foods are distinct from other types of dietary therapies as described below:
• Medical foods are specially formulated and processed; they do not exist in a natural food state.
• Medical foods must be labeled for the dietary management of a specific medical disorder, disease or condition for which there are distinctive nutritional requirements.
• Medical foods are intended to be used under medical supervision.
• Medical foods are intended only for patients receiving active and ongoing medical supervision.
While there is a compliance program, medical foods do not have to undergo pre-market review, registration or approval by the FDA. For osteoarthritis (specifically knee osteoarthritis), Limbrel (flavocoxid) is a popular prescription medical food.
About Limbrel
Brought to the U.S. market in 2004, Limbrel contains flavocoxid, a blend of concentrated flavonoids, primarily baicalin and catechin. Largely responsible for bright colored pigments in fruit and vegetables, flavonoids are found in virtually all plants. Primus Pharmaceuticals, Inc., the company which markets Limbrel in the U.S., claims that:
• The ingredients in Limbrel have both anti-inflammatory and antioxidant properties in a concentrated form which cannot be obtained through normal diet.
• Limbrel restores the body’s normal metabolic inflammatory processes through dual inhibition of excess arachidonic acid metabolization and antioxidant action.
• Limbrel manages production of arachidonic acid metabolites, while inhibiting COX-1, COX-2, and 5-LOX (5-lipoxygenase), all of which contribute to osteoarthritis.
Although it has not been around long enough to generate a long-standing safety profile, some doctors are finding Limbrel to be a safe and effective alternative to anti-inflammatory and pain medications for knee osteoarthritis. However, a recent study published in the Annals of Internal Medicine casts a caution on this medical food.
In a small study, Dr. Naga Chalasani, director of the division of gastroenterology and hepatology at Indiana University School of Medicine, and colleagues found that Limbrel was linked to several cases of liver disease. The authors concluded, “Our report provides convincing evidence that flavocoxid is capable of causing clinically apparent, acute liver injury.”
The incidence of liver problems in those taking Limbrel appears to be low, but those with preexisting liver disease might be more vulnerable than others to any additional stress on this organ. More information is needed to make an educated decision about whether or not those with liver disease should take Limbrel.
By integrating western medicine with nutrition, medical foods seem to fill an important role. However, Chalasani’s study is a reminder that just because it comes from a natural source, doesn’t mean it’s safe for everyone. People should not assume that medical foods (including Limbrel) are 100 percent safe. Because medical foods are not subjected to the clinical trials required of prescription drugs before coming to market in the U.S., caution is warranted. Currently, over 25,000 patients are being monitored in post-marketing surveillance associated with Limbrel.
There are, as described above, a long list of other treatment approaches to help manage osteoarthritis. Even so, those currently suffering from knee osteoarthritis may not want to wait for the final verdict on Limbrel to get the “close to natural” relief that this medical food has the potential to deliver.
http://arthritis.about.com/od/limbrel/a/limbrel.htm, Limbrel, Carol Eustice, Retrieved June 24, 2012, about.com, 2012.
http://health.usnews.com/health-news/news/articles/2012/06/18/arthritis-treatment-linked-to-liver-problems-in-study, Arthritis Treatment Linked to Liver Problems in Study, Retrieved June 24, 2012, US News & World Report LP, 2012.
http://osteoarthritis.about.com/od/kneeosteoarthritis/a/knee_OA.htm, Knee Osteoarthritis: What You Need to Know, Carol Eustice, Retrieved June 30, 2012, about.com, 2012.
http://www.fda.gov/food/foodsafety/product-specificinformation/medicalfoods/default.htm, Medical Foods, Retrieved June 24, 2012, US Food and Drug Administration, 2012.
http://www.fda.gov/Food/GuidanceComplianceRegulatoryInformation/GuidanceDocuments/MedicalFoods/ucm054048.htm, Guidance for Industry: Frequently Asked Questions About Medical Foods, Retrieved June 24, 2012, US Food and Drug Administration, 2012.
http://www.whfoods.com/genpage.php?tname=nutrient&dbid=119, Flavonoids, Retrieved June 30, 2012, The George Mateljan Foundation, 2012.






Hi. Is this medical food recommended for those osteoarthritis patients who were cured? I had OA and was totally healed through stem cell treatment (thanks to my ortho surgeon, Dr Purita). It has been 3 years and my joints are still working perfectly but I’m currently looking for foods and supplements that I need so I can maintain my joints in good shape. Thanks!
Most athletes might avoid these injuries, or at the terribly
least create them not as damaging, by employing a knee support to assist them
avoid this. A elegant brace is on the market that might facilitate any
contestant from pain themselves in a very lasting approach and may facilitate
to stay your season going, rather than depending on from (dare we are saying
it) “the bench”.
When a personal has water on the knee, pain relief will
occur once the fluid is off from the joint. Pain relief will be subsided once
this fluid returns.